New Neuroprotective and Accelerator of Nerve Regeneration Agent Described
12/12/2017
The research was directed by Caty Casas and included the participation of David Romeo and Xavier Navarro, researchers at the INc-UAB and CIBERNED, and Joaquim Forés of the Hospital Clínic Barcelona.
The new pharmacological agent NeuroHeal was designed to imitate and strengthen neuroprotective mechanisms which naturally command neurons to deal with minor injuries successfully.
The pre-clinical trials indicate that oral administration permits a long-lasting maintainance of the survival of damaged motor neurons, for at least six months, after peripheral nerve root avulsion, even in cases of delayed surgical reimplantation, as happens in clinical practices. In addition, the treatment accelerates nerve regeneration, drastically reduces denervation-induced muscular atrophy and increases functional contacts between the nerve and affected muscles.
“There is currently no pharmacological treatment indicated as an adjuvant therapy to maintain alive a neural population after such a severe injury and until surgical intervention, nor is there any to accelerate nerve regeneration. This new drug acts in both senses”, Caty Casas explains.
Injuries to peripheral nerves are consequence of traffic, work and sportive accidents that can cause nerve sectioning or compression. For example, a digital nerve section or injury in someone who works with dangerous machinery, or a high-energy impact to the shoulder in a motorcycle accident can produce nerve root avulsion in the most vulnerable areas, such as lumbar or brachial roots. The surgical reconstitution may be done days after the accident while the patient is stabilized and diagnosed but during that time, disconnected motor neurons due to nerve traction undergo a fatal and irreparable degenerative process.
In addition, if the roots and nerves are repaired through surgery, the surviving motor neurons must regenerate their axons through these same nerves until re-establishing a connection with the muscles they controlled and recovering movements. In this case it is also a race against time. It can take up to two years for patients to regenerate their nervous circuit. The longer it takes to regenerate, the more irreversible the muscle atrophy becomes, thus hindering a good recovery.
NeuroHeal is a Combination of Two Repurposed Drugs
NeuroHeal is a particular dose combination of two repurposed drugs, Acamprosate and Ribavirin, which are currently used to treat other unrelated diseases. Researchers designed it by using artificial intelligence-based computational tools and systems biology approaches to interpret available biological big data and simulate biological responses to thousands of pharmacological combinations. The combination to conform NeuroHeal presented the best profile amongst all of them. This computational study was conducted thanks to the collaboration with Anaxomics Biotech.
Researchers have already patented the new drug and hope that it will be used in the near future. “The fact that the two compounds in NeuroHeal are already being used and have demonstrated their pharmacokinetics and safety in humans, allow faster implementation for clinical use”, the researcher concludes.
Image:
http://www.uab.cat/uabdivulga/img/INc-UAB_NeuroHeal2.jpg
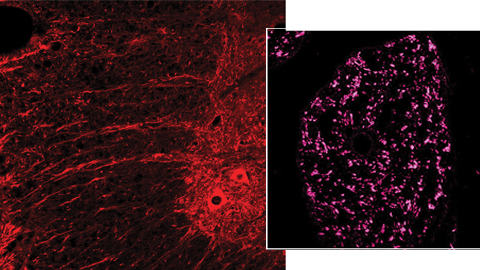
The analysis of the the spinal cord ventral horn and sciatic nerve (diagram indicating the areas) through confocal microscope clearly demonstrates that in the reimplantation model (RE), animals treated with NeuroHeal (NH) present more motor axonal ramifications (positive ChAT, in red) and more regenerated fibers (positive GAP43, in magenta) than untreated animals.
Original article: Romeo-Guitart D, Forés J, Navarro X, Casas C. Boosted Regeneration and Reduced Denervated Muscle Atrophy by NeuroHeal in a Pre-clinical Model of lumbar root avulsion with delayed reimplantation. Scientific Reports 7, Article number: 12028 (2017). DOI:10.1038/s41598-017-11086-3