New antitumoral drug release strategy created for breast cancer treatment
20/11/2019
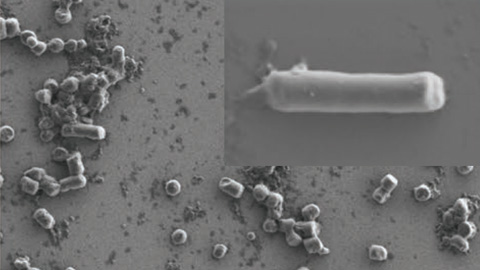
The local release of antitumoral drugs through bacterial proteins in the treatment of breast cancer can mark a before and after in precision medicine. In this sense, researchers at the CIBER of Bioengineering, Biomaterials and Nanomedicine (CIBER-BBN) of the Universitat Autònoma de Barcelona, the Vall d'Hebron Hospital Research Institute and the Spanish National Research Council (CSIC) have created Escherichia coli cell structures to produce non-toxic bacterial amyloids. Inclusion bodies or amyloids are nanostructured protein aggregates produced inside a cell, frequently found in specific bacteria and with interesting biomedical applications such as protein drug release, as is the case here.
These protein structures act as antitumoral drug secretion granules and since they are administered locally, they have a sustained therapeutic effect throughout time. Although this research, published in Advanced Science, is still in the first stages of development, the cross-cutting principle described in the study opens a broad experimentation field for the generation of new therapeutic biomaterials produced within bacteria for precision medicine aimed at breast cancer and other high incidence neoplasms.
This research project, funded by the TV3 Marathon Foundation, was coordinated by the UAB's CIBER-BBN researcher Esther Vázquez, and also included the participation of Ibane Abasolo (Vall d'Hebron Hospital) and Miriam Royo (CSIC). The consortium also included the collaboration of Laura Soucek from the Vall d'Hebron Cancer Research Institute (VHIO) and ICREA, and expert in animal models for the study of cancer.
According to Esther Vázquez, "although this technology still has a long way to go before being applied clinically, the results obtained in this study, which has lasted over three years, pave the way for a new therapeutic technology based on bacterial products which have not been clinically explored yet".
The study is based on a directed release of CD44+ tumour cells in animal models of human breast cancer, of two antitumoral proteins (Omomyc and p31) in the form of nanostructured materials. The main novelty of the study lies in the use of toxic-free bacterial amyloids as a reservoir for these therapeutic proteins. The local administration of this material fosters a sustained release of the drugs and the necrosis of tumour tissue in a relatively short period of time. One of the main advantages of using these types of materials is a sustained protein drug release, which lowers the frequency of administration in relation to the current standards for conventional drugs.
The project also included the collaboration of the lead researchers at the CIBER-BBN group Antonio Villaverde, from the UAB, expert in the development of bacterial materials of biotechnological interest based on amyloid structures, also known as inclusion bodies, and Simó Schwartz Jr, director of the CIBBIM-Nanomedicine (VHIR) and expert in nanomedicine and the molecular biology of cancer.
Original article:
Mireia Pesarrodona, Toni Jauset, Zamira V. Díaz‐Riascos, Alejandro Sánchez‐Chardi, Marie‐Eve Beaulieu, Joaquín Seras‐Franzoso, Laura Sánchez‐García, Ricardo Baltà‐Foix, Sandra Mancilla, Yolanda Fernández, Úrsula Rinas, Simó Schwartz Jr, Laura Soucek, Antonio Villaverde, Ibane Abasolo, Esther Vázquez. Targeting Antitumoral Proteins to Breast Cancer by Local Administration of Functional Inclusion Bodies, Advanced Science. https://doi.org/10.1002/advs.201900849