New article: "Conjugation strategy shapes antitumor efficacy and enables dose-sparing in non-antibody protein nanoconjugates"
Research Group: Nanobiotechnology
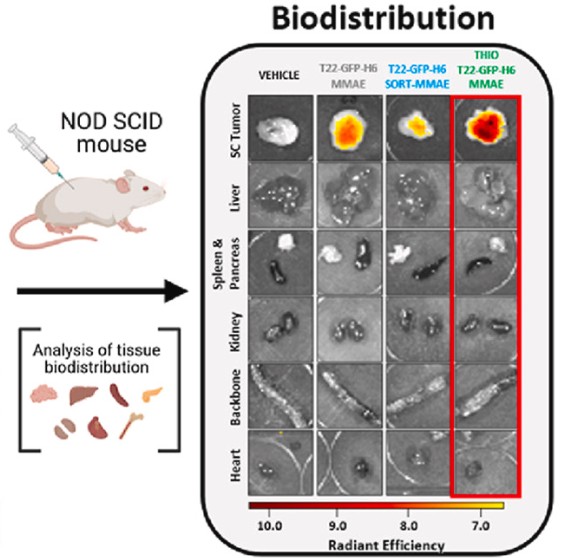
Abstract:
Precision targeting is a hot topic in cancer nanomedicine, as conventional chemotherapies cause systemic toxicities, creating an urgent need for more selective treatments. Although antibody-drug conjugates (ADCs) are the current gold standard in targeted therapy, their clinical performance remains limited. As an alternative, we previously developed a multivalent protein nanocarrier (T22-GFP-H6) displaying the CXCR4-targeting peptide T22, which offers super-selective tumor accumulation driven by CXCR4 overexpression. This innovative nanovehicle showed favorable biodistribution for targeted delivery of antitumor drugs, including monomethyl auristatin E (MMAE), in a first-generation stochastic nanoconjugate format. However, unlike ADCs, where conjugation strategy is known to influence pharmacokinetics and efficacy, these parameters remain largely unexplored in non-antibody multivalent nanocarriers. Here, we evaluated the impact of precise payload accommodation using two site-specific strategies that attach a single MMAE molecule at distinct structural sites, and we compared them with first-generation nanoconjugates. The conjugation strategy substantially affected the biodistribution and antitumor efficacy, with a solvent-exposed cysteine-conjugation distal to the targeting ligand proving most effective. At equimolar nanocarrier dosing, this construct achieved tumor control similar to the stochastic conjugate in a disseminated hematologic malignancy despite an approximately 4-fold lower MMAE load (drug-to-protein ratio, DPR = 1 vs DPR ≈ 4). Moreover, at equimolar MMAE dosing, it clearly outperformed both the stochastic conjugate and the alternative site-directed design. These findings align with trends in advanced ADCs and provide practical design rules for rational, site-specific conjugation in next-generation protein-based nanomedicines aimed at enabling dose-sparing in oncology.
Article data:
Ariana Rueda, Annabel Garcia-Leon, Lourdes A. Arena, Julian I. Mendoza, Anna Aviñó, Carme Fabrega, Ramon Eritja, David Paez, Lorena Alba-Castellon, Esther Vazquez, Antonio Villaverde, Ramon Mangues, Isolda Casanova, Ugutz Unzueta. Conjugation strategy shapes antitumor efficacy and enables dose-sparing in non-antibody protein nanoconjugates. Materials Today Bio, Volume 36, 2026,102698,ISSN 25900064. https://doi.org/10.1016/j.mtbio.2025.102698
The UAB, with the Sustainable Development Goals
-
Good health and well-being
