A new study from the ALBA Synchrotron explains the inefficacy of some diabetes drugs
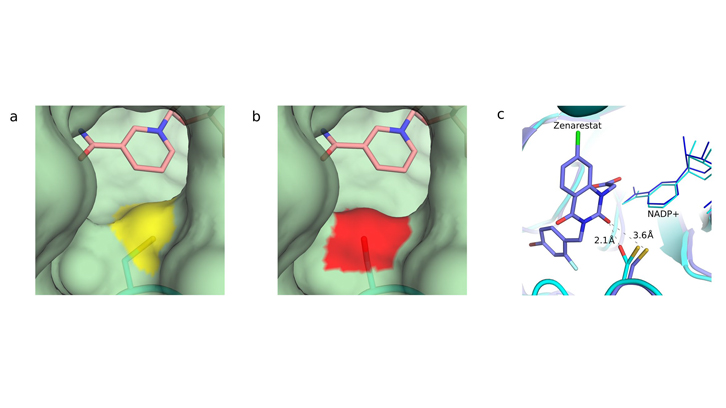
Figure 1. Changes on the aldose reductase pocket entry channel. The conversion of the cisteine into serine modifies the entrance of the substrate-binding pocket. a) Entrance of the binding pocket of the native form of aldose reductase. The sulfur atom of the cisteine is marked in yellow. b) Entrance of the binding pocket of the irradiated structure, which shows serine in red. In both cases, the nicotinamide ring of the cofactor involved is shown in pink color. c) Steric collisions of activated aldose reductase with one of the drugs tested: Zenarestat. The inhibitor, designed against the native form, show severe steric collisions with serine but not with cisteine.
The protein aldose reductase has been explored as a drug target since the 1980s for its implication in diabetic complications. Now, researchers Xavier Parés and Jaume Farrés, in collaboration with the team of the ALBA Synchrotron, have shown the reason why some drugs against the effects of diabetes under development do not work in the attempt to block aldose reductase. The results have been published in Scientific Reports Journal.
This protein has mainly detoxifying functions inside the cell but it can also transform glucose into a molecule called sorbitol. Under hyperglycemic conditions (high level of glucose in blood), this reaction increases much more and sorbitol accumulates, consuming antioxidant defenses. So, if hyperglycemia situation becomes chronic - like in diabetes -, there are unbalanced conditions inside the cell that lead to harmful oxidative stress environment.
To avoid this situation, it is necessary to obtain a drug able to inhibit the activity of aldose reductase to reduce the amount of sorbitol in the cell. The drug target is to bind to the protein union centre, taking up this part which corresponds to glucose. In this way, the chemical path that provokes oxidative stress would be stopped.
However, no effective drugs against aldose reductase have been developed yet. Although having good results in vitro, their potency is severely reduced in clinical trials. "The key point is that aldose reductase can appear under two different forms, the native and the activated one. Due to oxidative stress typical of diabetes, the protein suffers structural changes and the activated form comes out" explains Isidro Crespo, PhD student at ALBA. In spite of this change in the protein structure being known, drugs design is still focalized in the native form, instead of the activated one. The results obtained show in detail how the protein changes under the harmful effects of oxidative stress, and, more important, how these changes affect the efficacy of these drugs.
New method using synchrotron light
The XALOC beamline at the ALBA Synchrotron enables to determine the three-dimensional structure of the proteins. Moreover, this study takes advantage of the synchrotron light, formed basically by hard X-rays, to produce damage into the protein. "We have seen that, by irradiating the aldose reductase with synchrotron light, we can cause the same effects as those provoked in the protein under real oxidative stress at the diabetic patients cells," comments Albert Castellví, researcher of the team. "We managed to convert the native form of the protein into another that behaves like the activated one present in diabetic cells," he adds.
Once this activated form has been obtained, researchers have tested drugs that had already been rejected in clinical trials and has been confirmed that these inhibitors are not effective.
They conclude that it is due to the structural change that has suffered the protein after X-rays irradiation. Under oxidative stress conditions, the conversion of only one amino acid from the native protein (a cysteine) into other amino acids (mainly serine or alanine) affects the binding of the inhibitory drug to the protein and so, explains the changes in the drug’s efficacy. To get an effective drug, it must fit into the aldose reductase binding site as if it was a piece of a puzzle. Therefore, if the protein’s shape changes, the drug will not fit well into. For the researchers, this fact causes the inefficacy of drugs against diabetic complications and they propose a new alternative in the development of drugs: to design them so that they fit in the activated changed form of the protein, rather than the native one, to block it definitely.
Cellular oxidative stress cannot be reproduced in vitro due to the complexity and diversity of factors influencing this condition. "For this reason, the new method with synchrotron light can be so useful to simulate and study the changes that aldose reductase undergoes under oxidative stress conditions derived from diabetes," points out Judith Juanhuix, responsible researcher of this study. This method, in fact, could be extended to the study of other proteins that have suffered structural modifications due to oxidative stress and have a key role in diseases such as Parkinson, cancer or Alzheimer.
Universitat Autònoma de Barcelona
References
Castellví A, Crespo I, Crosas E, Cámara-Artigas A, Gavira JA, Aranda MAG, Parés X, Farrés J, Juanhuix J. (2019). Efficacy of aldose reductase inhibitors is affected by oxidative stress induced under X-ray irradiation. Sci Rep, 9(1):3177. DOI: 10.1038/s41598-019-39722-0.

