Effectiveness of a New Treatment for Atopic Dermatitis in Dogs
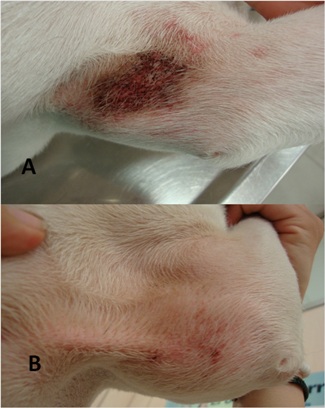
Atopic dermatitis, the second most common form of allergic dermatitis in dogs, has risen in the past decades. In fact, it is now thought that about 10% of dogs suffer from this condition. Nonetheless, the treatment options available are limited.Althoughallergen-specific immunotherapy remains the treatment of first choice because itis the safest way tomodulate the immune response, alternative treatments with immunosuppressive drugs, such as glucocorticoids or cyclosporine (CsA), are also available. The potent immunosuppressive activity of orally administered CsA is responsible for its efficacy but also for its toxic effects, related to the duration of treatment. Therefore, when a condition is chronic and CsA is used long-term, the potential risk increases.
Topical use of CsA has been hampered by its poor ability of absorption. Currently, nanotechnology has facilitated the development of a new pharmaceutical formulation able to penetrate through the epidermis to ensure absorption and dermal action of CsA.
The objective of this trial, conducted in multiples centres, was to evaluate blindly the efficacy of this new formulation of CsA in dogs with atopic dermatitis. Animals were randomly divided into two groups, one group treated with topical CsA and a control group treated with placebo. After 21, 30 and 45 days of topical CsA therapy all dogs showed an effective and significant reduction in pruritus and lesions severity. Conversely, the animals treated with placebo showed no significant improvement.
The results suggested that topical administration of CsA is effective in reducing the severity of skin lesions and pruritus in dogs with atopic dermatitis. Moreover, any of the animals treated with CsA experienced adverse events.
References
Puigdemont, A.; Brazís, P.; Ordeix, L.; Dalmau, A.; Fuertes, E.; Olivar, A.; Pérez, C.; Ravera, I. Efficacy of a new topical cyclosporine A formulation in the treatment of atopic dermatitis in dogs. Veterinary Journal 197(2): 280-285. 2013. DOI: 10.1016/j.tvjl.2013.02.018.

