Fluoro-tagged osmium and iridium nanoparticles in oxidation reactions
A few years ago, the use of highly fluorinated stabilizers for the preparation of metal nanoparticles did not seem appropriate. However, researchers from the UAB Chemistry Department showed the possibility of preparing and stabilizing metallic nanoparticles, in particular iridium and osmium, using a highly fluorinated stabilizer compound. In addition, once synthesized, they studied its applicability in oxidation reactions as well.
Metal nanoparticles (Mnp) have been subject of study for their unusual properties derived from their size. Normally, its preparation is carried out by chemical, electrochemical or thermal decomposition of a metal salt. Among the different preparation methods, the common point is to use a stabilizer to avoid agglomeration. A few years ago, the use of highly fluorinated compounds as stabilizers did not seem appropriate, but our research group in collaboration with others showed that it was possible to use stabilizers based on the presence of long perfluorinated chains.
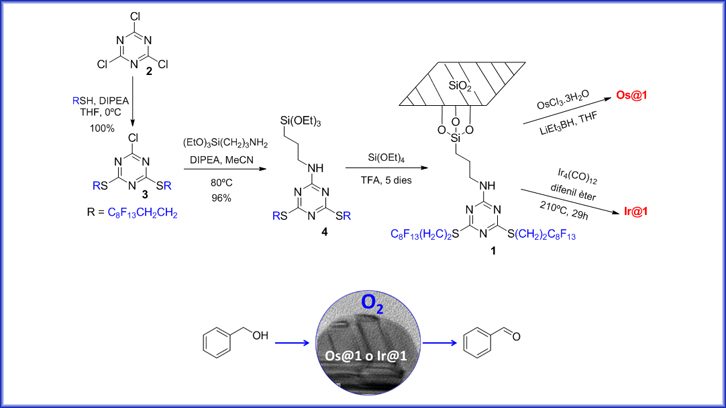
In this work, we have prepared metal iridium and osmium nanoparticles stabilized by a highly fluorinated organic-inorganic hybrid (1) and we have studied their applicability in the aerobic oxidation reactions of organic compounds (Scheme 1). The stabilization is based on the interactions of the Mnp with the nitrogen’s of the triazine nucleus and the polyfluorinated chains, which with their star-like disposition avoid the Mnp agglomeration.
The preparation of 1 is based on the reaction of 2,4,6-trichloro-1,3,5-triazine (2) with two equivalents of 1H,1H,2H,2H-perfluorodecantiol at 0ºC, by an aromatic nucleophilic substitution (100% yield). The chlorine remaining in the structure is replaced by the indicated silylated amine (Scheme 1), giving rise to the triazine (4) in 96% yield. The gelification of 4 gives rise to material 1 using trifluoroacetic acid and large excess of tetraethoxysilane.
Material 1 was used as a stabilizer in the preparation of osmium nanoparticles (Os@1) by reducing OsCl3•3H2O with LiEt3BH (Scheme 1). The Os@1 obtained were uniform, with good dispersion and a size of 1.3 ± 0.2 nm. The measurements by X-ray diffraction (pXRD) allowed to identify the compact hexagonal structure of Os(0). The formation of iridium nanoparticles was then investigated. It is important to mention that most of the reported syntheses of this kind of nanoparticles involve the reduction of an iridium salt by a reducing agent in the presence of a good stabilizer. Since this methodology did not give us good results, the preparation of nanoparticles by thermolysis caught our attention. Thus, the thermolysis of the cluster dodecarbonyltetrairidium (Ir4(CO)12) was carried out in diphenyl ether as solvent and in the presence of 1, yielding Ir@1 and CO (Scheme 1). The reaction could be monitored by infrared spectroscopy, since the absorption of the carbonyl group was not observed upon the completion of the reaction. The Ir@1 had an average size between 0.5-1.4 nm of diameter and the pXRD showed the faced centered cube structure of Ir(0).
Once the two new materials (Os@1 and Ir@1) were prepared, we explored their capacity to promote the aerobic oxidation of several benzyl alcohols to benzaldehydes. The experiments were carried out in toluene and under an O2 atmosphere at 80ºC. In the case of benzaldehyde, 85% yield was achieved when 2% Os@1 was used, at 100ºC and 2.5 atmospheres of oxygen pressure. When we tested the Ir@1 in the same oxidation reaction, it was observed that the material had to be pre-activated with heat (135ºC) for 2 hours to have good activity, mainly due to the presence of residual CO on the Ir@1. Under these conditions, benzaldehyde was obtained with a 97% yield. In any of the cases oxidation to the acid was observed.
Adelina Vallribera i Albert Granados
Departament of Chemistry
Universitat Autònoma de Barcelona
Universitat Autònoma de Barcelona
References
Santacruz, L, Donnici, S, Granados, A, Shafir, A & Vallribera, A. (2018). Fluoro-tagged osmium and iridium nanoparticles in oxidation reactions. Tetrahedron, 74 (48), 6890-6895. DOI: 10.1016/j.tet.2018.10.040.

