Computational methods help finding drug binding sites on cellular receptors
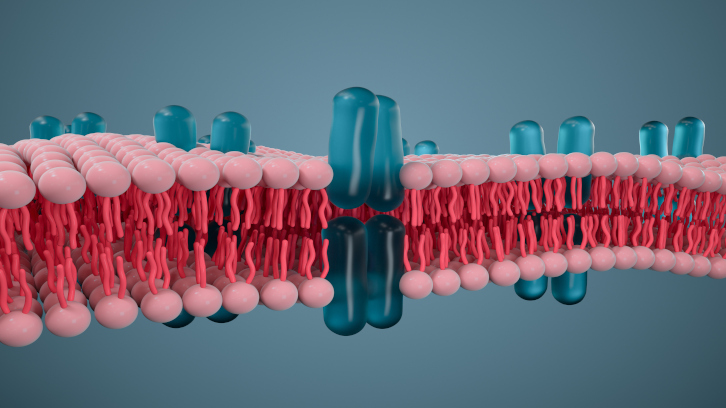
The dysfunction of G protein-coupled receptors, known as GPCRs, is associated with neurological, cardiac, and psychiatric disorders, among others. Drugs that target a specific GPCR can cause unwanted effects, since they usually bind to a site in the receptor which is quite evolutionarily conserved; therefore these drugs may end up interacting with other, distinct from the original target. In this article, the research group led by Dr. Jesús Giraldo from the Institute of Neurosciences proposes the use of computational models to search for other, less conserved drug binding sites in these receptors. The results, which will need to be verified experimentally, reinforce the role of computation as an ally for research in this field.
On the surface of our cells there are proteins called receptors, which are able to detect a signal from the surrounding environment and send this message into the cell. Among them, a very special group is formed by G Protein-Coupled Receptors, or GPCRs. GPCRs traverse the cell membrane: one end of the receptor faces outward (the extracellular environment) and the other faces the inside of the cell. These receptors are activated when a molecule binds to the receptor's cavity facing the extracellular medium — the orthosteric site — leading to a conformational change that opens a cavity at the other end, favoring the interaction of the receptor with molecules inside the cell, especially a signaling molecule called G Protein. This activation leads to a complex cellular response to the signal coming from outside.
Scheme: Arrangement of the GPCR receptor in the cell membrane and the orthosteric site
GPCRs are involved in virtually all physiological processes, including the senses of sight, taste and smell. The malfunction of one of these receptors can lead to neurological, cardiac or psychiatric disorders, among others. Consequently, they are very important therapeutic targets and it is estimated that about 30% of the drugs on the market act on GPCRs. Usually, these drugs regulate the activation of the receptor by binding to the orthosteric site, but this has disadvantages: these sites tend to be more conserved, at evolutionary level, among different GPCRs, so that a drug aimed at a specific receptor may end up interacting with another, causing unwanted effects. A possible alternative is to direct drugs to other cavities, called allosteric sites. They tend to be less conserved than orthosteric sites and therefore offer the possibility of greater specificity.
To explore these sites, first we need to find them: we must map the cavities and check which ones have the greatest potential to interfere with the functioning of the receptor when occupied by a drug. This was the objective of the work developed by us at the Laboratory of Molecular Neuropharmacology and Bioinformatics at the Institute of Neuroscience, coordinated by Dr. Jesús Giraldo. We performed a computational study in which we evaluated the extent to which the internal geometry of cavities on the surface of GPCRs accompanies the global conformational change of the receptor in the activation process. We examined structures obtained by X-ray crystallography and also simulations that generate different conformations of a receptor, and found that cavities whose geometry is more strongly correlated to the global dynamics of the receptor correspond to experimentally known allosteric sites. Our results suggest that these correlations between the local dynamics of the cavities and the collective dynamics of the receptor may help to predict the location of allosteric sites.
Figure 1 Coupling between the local dynamics of cavities in a GPCR and the collective dynamics of the receptor. Cavities are represented by spheres, which are colored according to the coupling strength (from blue, denoting weaker coupling, to red, indicating stronger coupling). Visit the original article to see the full image
Our hypothesis must now be tested by experiments, which will tell whether our method does indeed have predictive value. In case of success, we will have at our disposal one more tool to join efforts in the search for more effective and safer drugs.
*This project has received funding from the European Union’s Horizon 2020 research and
innovation programme under grant agreement N. 848068
Pedro Renault and Jesús Giraldo
Laboratory of Molecular Neuropharmacology and Bioinformatics, Unitat de Bioestadística and Institut de Neurociències, Universitat Autònoma de Barcelona.
Unitat de Neurociència Traslacional, Parc Taulí Hospital Universitari, Institut d’Investigació i Innovació Parc Taulí (I3PT), Institut de Neurociències, Universitat Autònoma de Barcelona.
Instituto de Salud Carlos III, Centro de Investigación Biomédica en Red de Salud Mental (CIBERSAM).
References
Renault, P., and Giraldo, J. 2021. Dynamical Correlations Reveal Allosteric Sites in G Protein-Coupled Receptors. International Journal of Molecular Sciences 22(1), 187; https://doi.org/10.3390/ijms22010187

