Advanced image processing techniques for automatic detection of polyps, colorectal cancer precursor lesions.
Colorectal cancer is one of the diseases with the highest impact worldwide, with more than 40,000 new cases annually only at national level. Its diagnosis is performed from the identification of its precursor lesion, the polyp. Polyps are benign in their early stages, but they can develop into cancer over time. Therefore, early detection of these lesions is key to ensure patient survival.
During the explorations, clinicians explore colon walls searching for polyps; although there are alternative techniques such as wireless capsule endoscopy or virtual colonoscopy, still colonoscopy is gold standard tool for colon screening as it allows lesion detection and removal for histological analysis during the same. Despite this, some polyps are not found during explorations, especially those small/flat or those hidden in the colon folds. In order to overcome this, several areas of improvement have been proposed, from the development of more advanced visualization systems allowing better lesion identification to the development of computational support systems assisting the clinicians in the different stages of a colonoscopy intervention. The objective of our research is the design of these computational support systems. Our developments have been carried out jointly by researchers from the Computer Vision Center (CVC) and the Digestive Endoscopy Unit of Hospital Clinic of Barcelona.
The computational support system that we propose is based on the use of image processing techniques for the automatic detection of polyps and the posterior characterization of polyp surface, once the area that occupies the polyp has been correctly delimited, to determine its degree of malignancy.
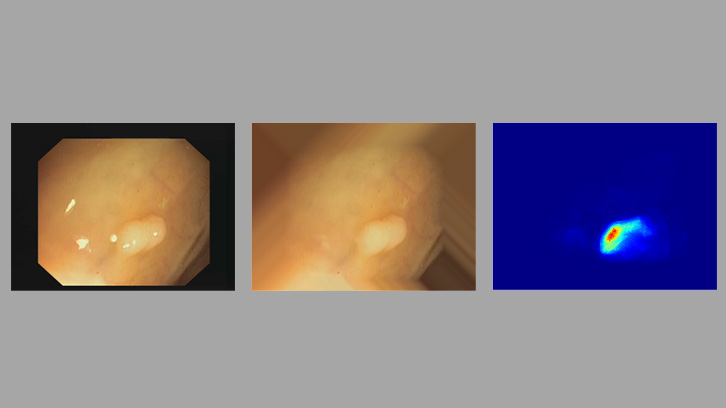
Our methodology for the automatic polyp detection is based on the definition of a model of polyp appearance that defines them as structures protruding from the colon wall. These structures appear surrounded by shadows when illuminated perpendicularly by the endoscope therefore, the automatic identification of these shadows can help the detection of polyps. Our method is based on the identification of these shadows as intensity valleys. Our system smartly integrates information from these intensity valleys to highlight those areas of the image more likely to contain a polyp. This valley information is also used to delimit the region that occupies the polyp in the image.
Our polyp detection system has been validated in several images and videos provided by the Digestive Endoscopy Unit of Hospital Clinic de Barcelona. Our method is able to detect all lesions that appear in the different databases, showing especially good performance effectiveness for flat lesions, which were identified by clinicians as the most challenging ones. In addition, we have compared the identification of areas of interest provided by our method with those provided by clinicians for the same exploratory studies; our results show similar pattern search behaviors between our system and clinical staff.
These results show the potential of our polyp detection system to be used in the exploration room to effectively support clinical staff.
To extend information, link to informational video:
http://www.cvc.uab.es/CVC-Colon/index.php/apodec/
jorge.bernal@uab.cat
Department of Computer Science
Universitat Autònoma de Barcelona
References
Bernal, J., Sánchez, J., & Vilarino, F. (2012). Towards automatic polyp detection with a polyp appearance model. Pattern Recognition, 45(9), 3166-3182. DOI: https://doi.org/10.1016/j.patcog.2012.03.002
Bernal, J., Sánchez, F. J., Fernández-Esparrach, G., Gil, D., Rodríguez, C., & Vilariño, F. (2015). WM-DOVA maps for accurate polyp highlighting in colonoscopy: Validation vs. saliency maps from physicians. Computerized Medical Imaging and Graphics, 43, 99-111. DOI: https://doi.org/10.1016/j.compmedimag.2015.02.007
Fernández-Esparrach, G., Bernal, J., López-Cerón, M., Córdova, H., Sánchez-Montes, C., de Miguel, C. R., & Sánchez, F. J. (2016). Exploring the clinical potential of an automatic colonic polyp detection method based on the creation of energy maps. Endoscopy, 48(09), 837-842. DOI: https://doi.org/10.1055/s-0042-108434


